Methodology:
- Setting the pioreactor and experiment environment (no media exchange during experiment)
- Ed01: 4%;no chloride media; initial OD600 0.05 CH34
- Ed02: 3.5%; no chloride media; initial OD600 0.05 CH34
- Ed03: 3%; no chloride media; initial OD600 0.05 CH34
- Incubator 1: no electrolysis; no chloride media; initial OD600 0.05 CH34
- Incubator 2: no electrolysis; no chloride media + 0.01% Yeast Extract; initial OD600 0.05 CH34
- After 48 hour incubation at 30C, adding sample with series dilution on the LB agar without antibiotic to test CFU at 30C incubator
- After 24 hour, the colonies are not formed
- After another 24 hours, the colonies are formed and counted
Results and discussion:
-
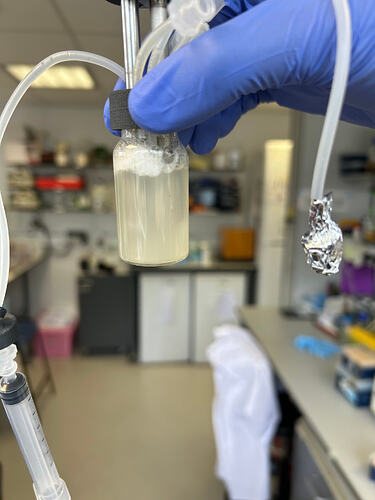
Potential biofilm forming on cathode shown in Figure 1. After removing the caps with electrodes from the vial, I discovered that something crystal and a little white attaching to the cathode. The reason that I say attaching is that it cannot be removed by wahsing with water. THerefore, I think this is not any crystallization of ions from the media.
Figure 1, the possible biofilm on cathode. -
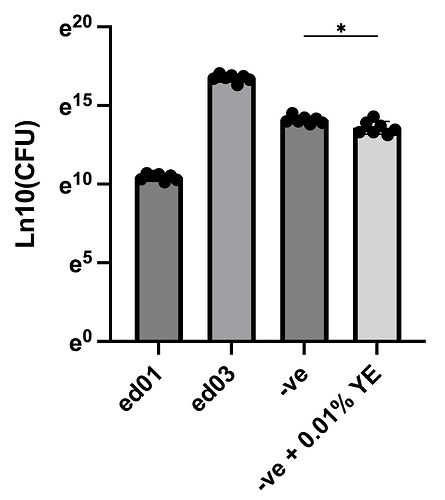
Electrolysis above 3% cannot support growth but it seems that it can main the survival. This conclusion is based on one phenomenon and the CFU results. According to Figure 2, the ed02 (3.5%) cannot support any obvious growth after 48 hour incubation. However, the figure 3 showing ed03 (3%) support the growth very well (I do not know why there are lots of bubbles). The CFU test is for trial so I only applied 10ul drop on agar instead of spreading to the whole agar. According to the results in Figure 4, the ed03 (3%) shows about 1000X survival than ed01 (4%) and 100-500X survival than ed02 (3.5%). Interestingly, ed03(3%) even show10X survival than CH34 incubating in incubator. Finally, 0.01% yeast extract does differ the survival of CH34 in incubator at 30C for 2 days according to the CFU test, and media without YE show good survivial supreisingy, but it is not very persuasive.
Figure 2, ed02 (3.5%) after 48 hour growth at 30C
Figure 3, ed03 (3%) growth after 48 hour under 30C
Figure 4, the CFU test (ed01 and ed03 is significantly different, not shown here). -
Anode is not black and without metal bluster under 4% electorlysis but some black things covered the whole cathode, shown in Figure 5. First, the covering happened to all the cathode instead of the part facing to the anode. Second, the black things can be partially wiped with 70% ethanol or partially disappear under 70% ethanol submerge but cannot be erased totally.
Figure 5, the black covering ed01 cathodes. The part submerged in the media is black.
Discussion:
- the biofilm appeared in hydrogen appearing electodes has been reported before, but we need to test wheter that is CH34 biofilm or contamination or some crystalliation. After submergin in 70% ethanol for 2 days, the attach things is still like the cotton or connective tissue in human body, so I suggest it might be biofilm.
- I am totally sure that electrolysis happened in ed03, but its incredibly good growth and survival surprise me. Because the experiment lack so many control, like the potential test, no constitutive test, just one setting wihtout replication, I suggest to repeat the experiments with more control so we can acquire more precise and concise data. CFU test may be counted after 36 hours or 30 hours after adding sample to the agar
- To avoid the contamination, I will test whether Kanamycin and Spectinomycin will be dysfunctional after electrolysis. I pick these two with two points: 1. CH34 cannot be killed by these two in LB but I am not sure whether these two will slow the growth. 2. These two antibiotics does not possess chloride in their chemcial structure. I think the media with some antibiotic can really protect from contamination but cannot avoid totally.
Other things report:
-
I will make autoclave stock of different chemical in meso-nutrient, so it is more effective and safter than filtering everything together. Also, high temperature in autoclaving can help to dissolve. Iron concentration will not change, but H2PO4 will decrease to Lisa’s recipe.
-
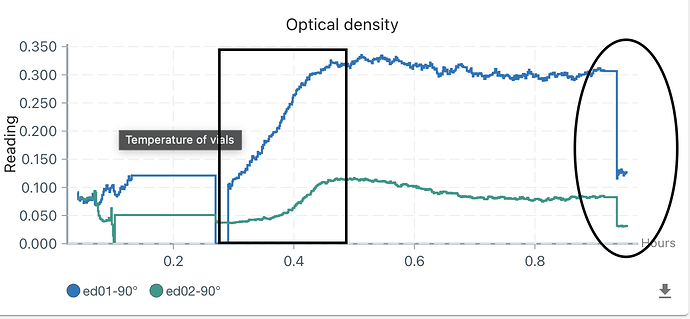
OD reading is really affected by the electrolysis shown in figure 6. The square showed the starting of electrolysis, and the circle showed the shutting down of electrolysis.
Figure 6, the OD reading affected by electrolysis. Square means starting electrolysis and circle indicated stoppingl. -
The media and product bottle cannot make sure the solution in that is sterile. After putting the media in the media bottle for 3-5 days, all of them are contraminated in three pioreactors.