My recent work utilised meso-nutrient, iron, potassium acetate, and acidified trace elements, but these do not support growth in our pioreactor. It happened to all of my reactors, but it worked in my flask in the incubator. However, I think a horrible incident happened at the reactor when I left, and I will explain it in detail.
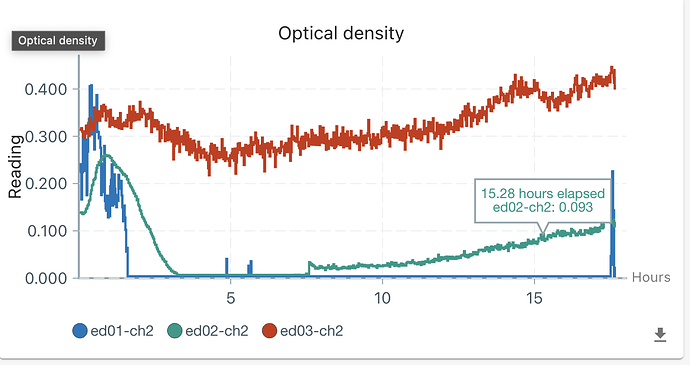
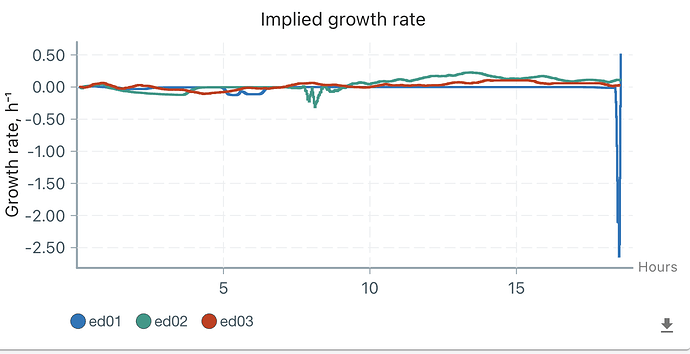
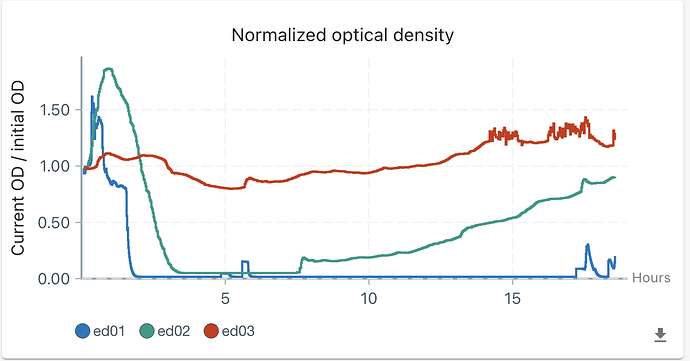
First, it is about media . The growth (OD) supervision started when I added the bacteria solution, so the initial OD is 0.2-0.3 (my calibration is not quite accurate as the model is not quite fitting). Then, the CH34 grow in the first 2 hours in ed01 and 02 and a little in ed03 (this calibration is not precise, and I will say that later). However, the OD decreased sharply to 0 after 2 hours. Although I set turbiostatic (target OD = 0.4), ed01 and ed02 do not activate overnight, which means the OD should only increase if no clear media was pumped in and our media worked. I measured OD on our machine this morning, and it’s still under 0.25, so I suggested nothing is growing. Therefore, I can only say our media does not support CH34 growth.
Second, the weird re-growth in ed02. I do not know why OD increased 5 hours after OD decreased to 0. Instead of ed01, whose curve stays at 0 (the final peak is the time I use the needle to take some solution). Therefore, I might think that is contamination, but two phenomena cannot be explained. First, the OD does not change at the point of adding bacteria yesterday and final measuring just now. If there is contamination, the OD should increase. Second, the curve is too smooth to be attributed by contamination.
Third, the OD calibration remains imprecise at higher ODs. Indeed, I find some difficult things when I calibrate OD. First, the needle will touch the electrodes and the CO2 tubes when injecting and sucking out media, which might affect the calibration and official experiments. Second, we probably need to find something to seal the hole for adding or taking out solution (i use tape to seal).
Therefore, there are multiple strange things that we need to discuss.
- Why does OD go down after a boost? I think this is an OD measurement issue, since the OD at the start and end points remains the same.
- What happened to ed02?
- I need to retest our media with acidified trace elements again in the incubator and pioreactors overnight, compared with un-acidified trace elements. My pH is lower than Imperial’s, so if mine does not work very well, theirs may show worse results.
- Need a whole protocol from receiving autoclaved vials to starting experiments (making media, bottle assembly, calibrations, and final bacteria addition). We need this to minimise the contamination and ensure more precise calibrations.